Agentic Development Life Cycle (ADLC) Management Process
Agentic Development is an emerging approach to application development that leverages autonomous AI agents. The complexity and self-directing nature of these agents necessitate a robust and ethical management process. Cyli Solutions LLC is establishing an Agentic Development Life Cycle (ADLC) process, borrowing from the highly regulated and rigorous clinical trial process of the pharmaceutical industry to ensure ethics, security, safety, and compliance.
Yong Li
3/17/20261 min read
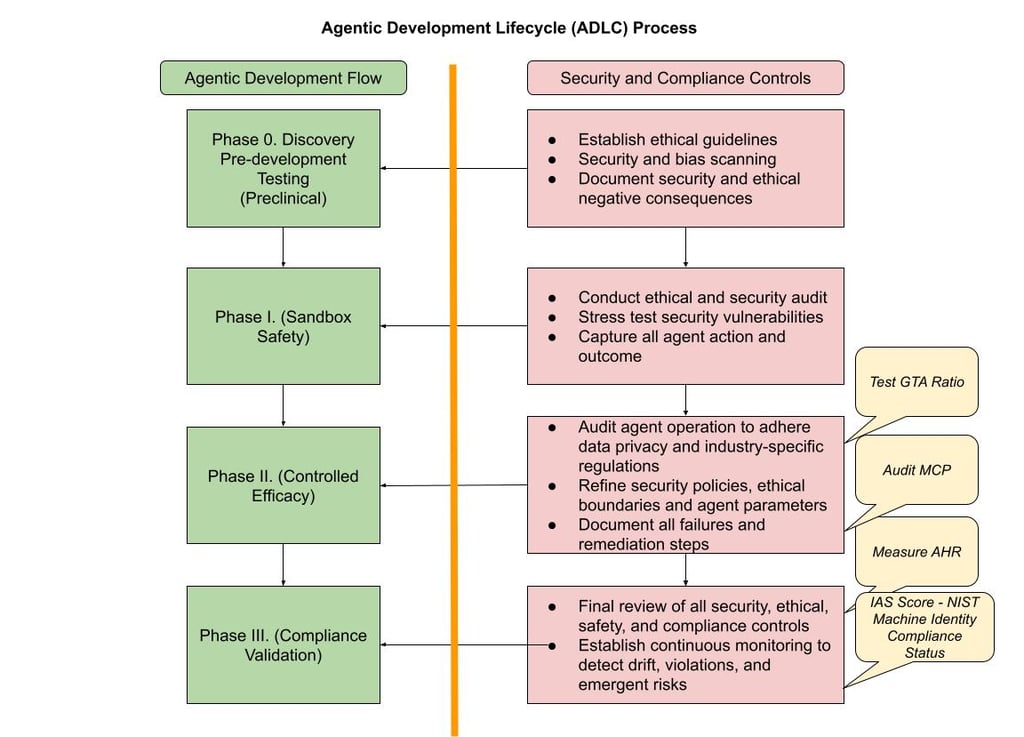
Stages of the Agentic Development Life Cycle (ADLC)
The ADLC mirrors the four phases of pharmaceutical clinical trials, ensuring agent maturity and safety before general deployment.
Phase 0: Pre-clinical Development (Discovery and In-Vitro Testing)
This stage is for initial concept validation and basic safety assessment in a controlled environment.
Phase I: Internal Validation (Safety and Dosing)
Focus on safety and feasibility with small, internal, non-critical use cases.
Phase II: Limited Pilot (Efficacy and Side Effects)
Test the agent's effectiveness and measure unintended consequences in a controlled external setting.
Phase III: General Readiness (Confirmation and Large-Scale Testing)
Final stage before full production deployment, focused on reliability and consistency at scale.